Most Chemical Reactions Can Be Divided Into Five Main Groups
Organic cofactors are sometimes further divided into coenzymes and prosthetic groups. The partially filled d sublevel makes it a member of the d-block.
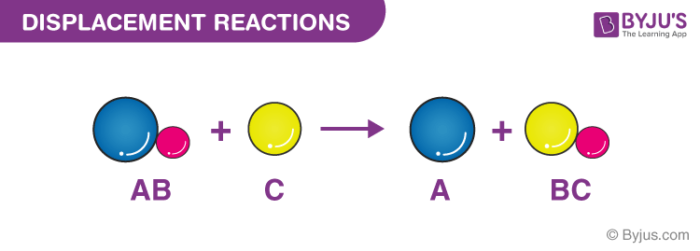
Displacement Reactions Definition Types Single Double Examples
Minerals are classified based on their crystal form and chemistry.

. Oxidoreductases transferases and hydrolases. True the five groups are combustion synthesis decompostion single displacement and double discplacement. Elements are substances that cannot be broken down into simpler ones.
Most plastics are poor conductors of heat. A constant new supply of red blood cells is needed to replace old cells that break down. The process works by heating DNA so that it separates into two single strands.
So elements can exist in the form of ions atoms isotopes molecules. 1 synthetic chemicals 2 chemical substances or metabolic products made by microorganisms and 3 chemical substances derived from plants. The term coenzyme refers specifically to enzymes and as such to the functional properties of a protein.
Examples of metallic minerals. Examples of Elements Elements exist in their simplest form and cannot be broken down further. They mostly incorporate metals nonmetals or metalloids.
The catalytic action of these enzymes may be reproduced by means of trivalent iron ions. The 5 primary types of chemical reactions are. And inorganic cofactors such as the metal ions Mg 2 Cu Mn 2 and ironsulfur clusters.
Metallic minerals exhibit lustre in their appearance and consist of metals in their chemical composition. The monosaccharides can be divided into groups based on the number of carbon atoms in the molecules thus. Organic cofactors such as flavin or heme.
Combination Reaction A reaction in which two or more reactants combine to form a single product is known as a combination reaction. It takes the form of X Y XY. Most chemical processes can be studied directly in a laboratory using a.
Polymerase chain reaction PCR is by far the most complicated reaction on this list but potentially the most useful and exciting. The composition statistical properties transformations and reactions of these materials are studied. Chemistry also involves understanding the properties and interactions of individual atoms and molecules for use in larger-scale applications.
By adding 6 2 8 we can place the element in Group 8. The element is in Period 6. Millions of red blood cells are made each day.
Within each of these groups there are different compounds each with the same molecular formula. The carbonyl group CO is probably the most important functional group in organic chemistry. The element is osmium a metal specifically a transition metal.
One drop of blood contains about five million red cells. Cofactors can be divided into two major groups. Combination reaction Decomposition reaction Displacement reaction Double Displacement reaction Precipitation Reaction 1.
These are again further divided depending upon their properties. Minerals are divided into two types namely metallic and non-metallic. Those involve carbon dioxide carbon monoxide cyanates cyanides and other carbon-containing ion compounds.
As discussed above during gravimetric analysis an excess of added reagent reacts with the analyte to form a precipitate. These compounds are an integral part of organic chemistry and their primary members are called aldehydes ketones and carboxylic acids. Trioses have 3-C atoms tetroses have 4-C atoms pentoses have 5-C atoms and hexoses have 6-C atoms.
These agents generally are of three types. Grain standards includes i breakage susceptibility ii milling quality iii seed viability iv nutritive value v mold count and carcinogen content and vi insect infestation. As an example both glucose and fructose are hexoses C.
According to the type of reactions that the enzymes catalyze enzymes are classified into seven categories which are oxidoreductases transferases hydrolases lyases isomerases ligases and translocases. There are a few carbon compounds which are. These substances are divided into elements and compounds further.
For instance foamed polystyrene used in cups for hot beverages has a thermal conductivity about one-quarter that of the unfoamed polymer. The carbon atoms join together to form the framework of the compound and the hydrogen atoms attach to them in many different configurations. Natural science can be divided into two main branches.
Blood cells are divided into three main types. These make blood a red colour. These can be seen under a microscope and make up about 40 of the bloods volume.
The chemical industry comprises the companies that produce industrial chemicalsCentral to the modern world economy it converts raw materials oil natural gas air water metals and minerals into more than 70000 different productsThe plastics industry contains some overlap as some chemical companies produce plastics as well as chemicals. Both methods utilize exhaustive chemical reactions between the analyte and added reagents. An element cannot be transformed into a new element even by using any physical or chemical means.
Classical quantitative analysis can be divided into gravimetric analysis and volumetric analysis. Non-grain-standards properties edit Important non-grain standards in US. They serve as fuels.
The main groups are numbered from 1 to 7 going from left to right and the last group on the right is Group 0 the block in between Group 2 and Group 3 is where the transition metals are placed. These minerals serve as a potential source of metal and can be extracted through mining. Hydrocarbons are the principal constituents of petroleum and natural gas.
It has 2 valence electrons. Hydrocarbon any of a class of organic chemical compounds composed only of the elements carbon C and hydrogen H. PCR was invented in 1983 by Kary Mullis who eventually won a Nobel Prize for his work.
The compounds containing a carbonyl group the -CO group are called carbonyl compounds. Other elements such as phosphorus nitrogen and oxygen are also sometimes bound to carbon. Catalase catalyzes the reaction of the breakdown of hydrogen peroxide into water and oxygen and peroxidase uses peroxides to oxidize certain compounds for example polyphenols with the ensuing formation of homologous quinone and water.
In the grading systems or specification damaged kernels is divided into two main parts i heat damaged and ii total damaged. Conductivity can be reduced even further by incorporating a gas usually air into the material. Metallic is further classified into Main Group Metals Transition Metals and f-block metals.
In determining the group the f electrons can be ignored since they do not affect groups. Plastics also are electrical insulators unless especially designed for conductivity. On the other hand compounds are also pure substances formed.

Chapter 6 Quantities In Chemical Reactions Chemistry

5 3 Types Of Chemical Reactions Chemistry Libretexts
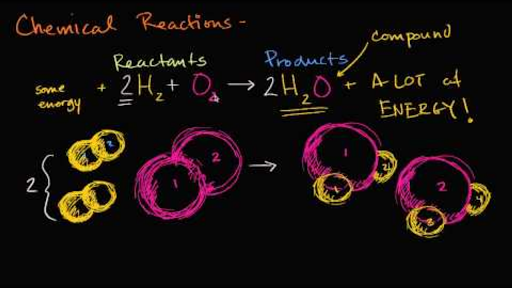
Chemical Reactions Introduction Video Khan Academy
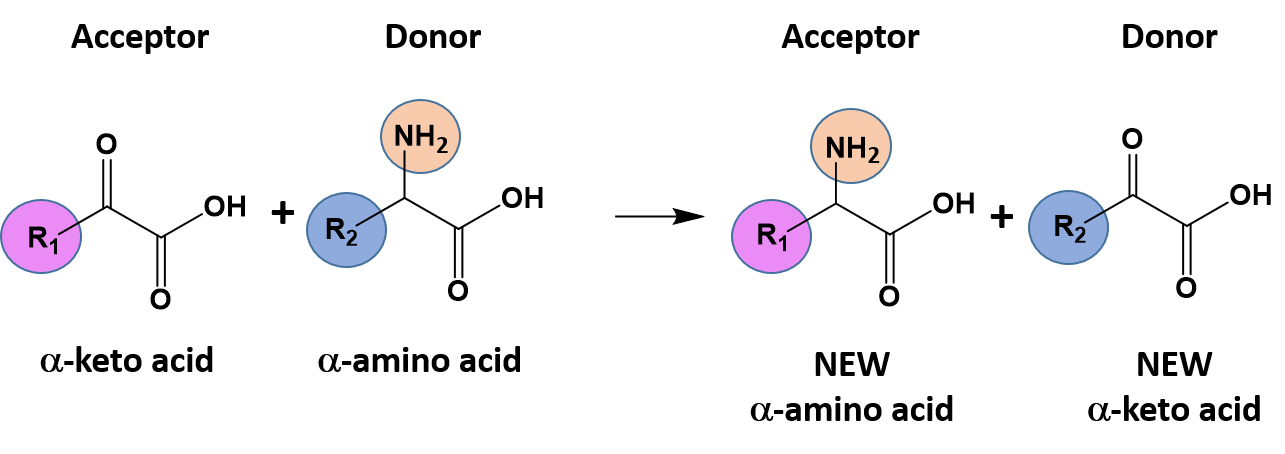
Ch103 Chapter 7 Chemical Reactions In Biological Systems Chemistry

Chemical Reactions Introduction Video Khan Academy
Chemical Reactions Chemistry Of Life Biology Article Khan Academy

Types Of Chemical Reactions Graphic Organizer Foldable With Dichotomy Key Educents Graphic Organizers Chemical Reactions Physical Science

Lakhmir Singh Chemistry Class 10 Solutions For Chapter 1 Chemical Reactions And Equations Free Pdf
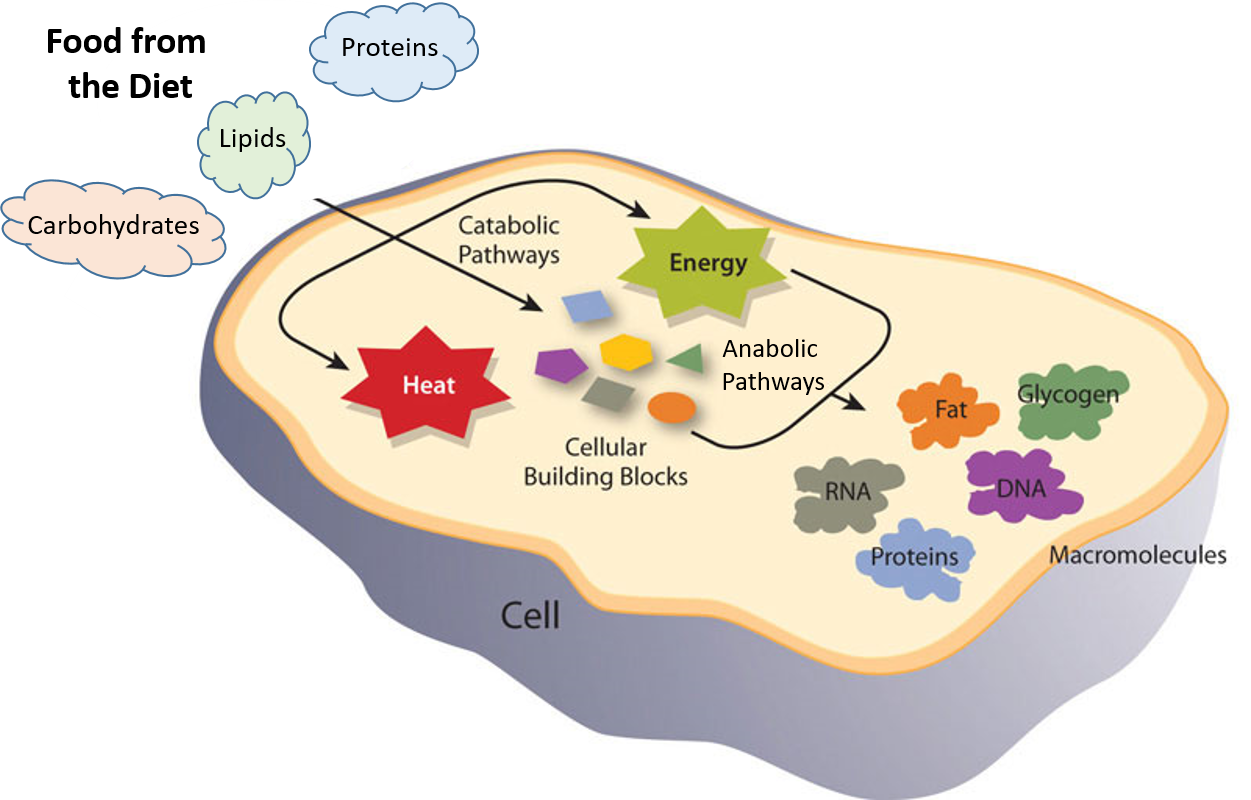
Ch103 Chapter 7 Chemical Reactions In Biological Systems Chemistry
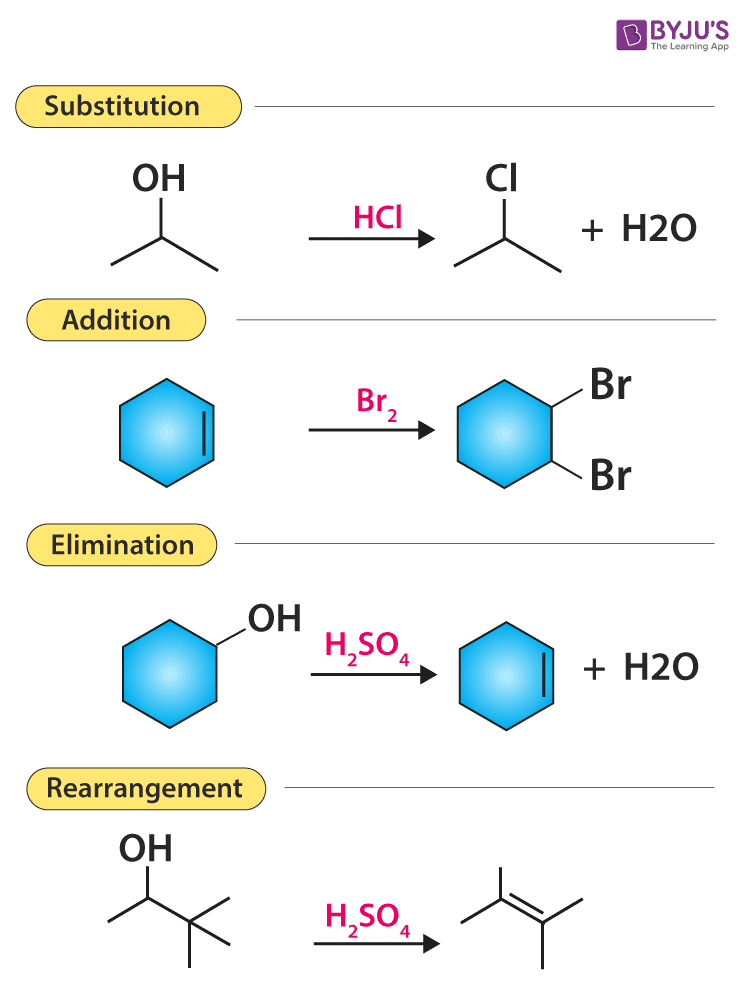
Various Types Of Organic Reactions Polar And Radical Reaction

Chemical Reactions Combination Decomposition Combustion Single Double Displacement Chemistry Youtube

Summary Poster On Vsepr Read More Here Millions Of Unique Designs By Independent Artists Find You Chemistry Classroom Chemistry Lessons Teaching Chemistry
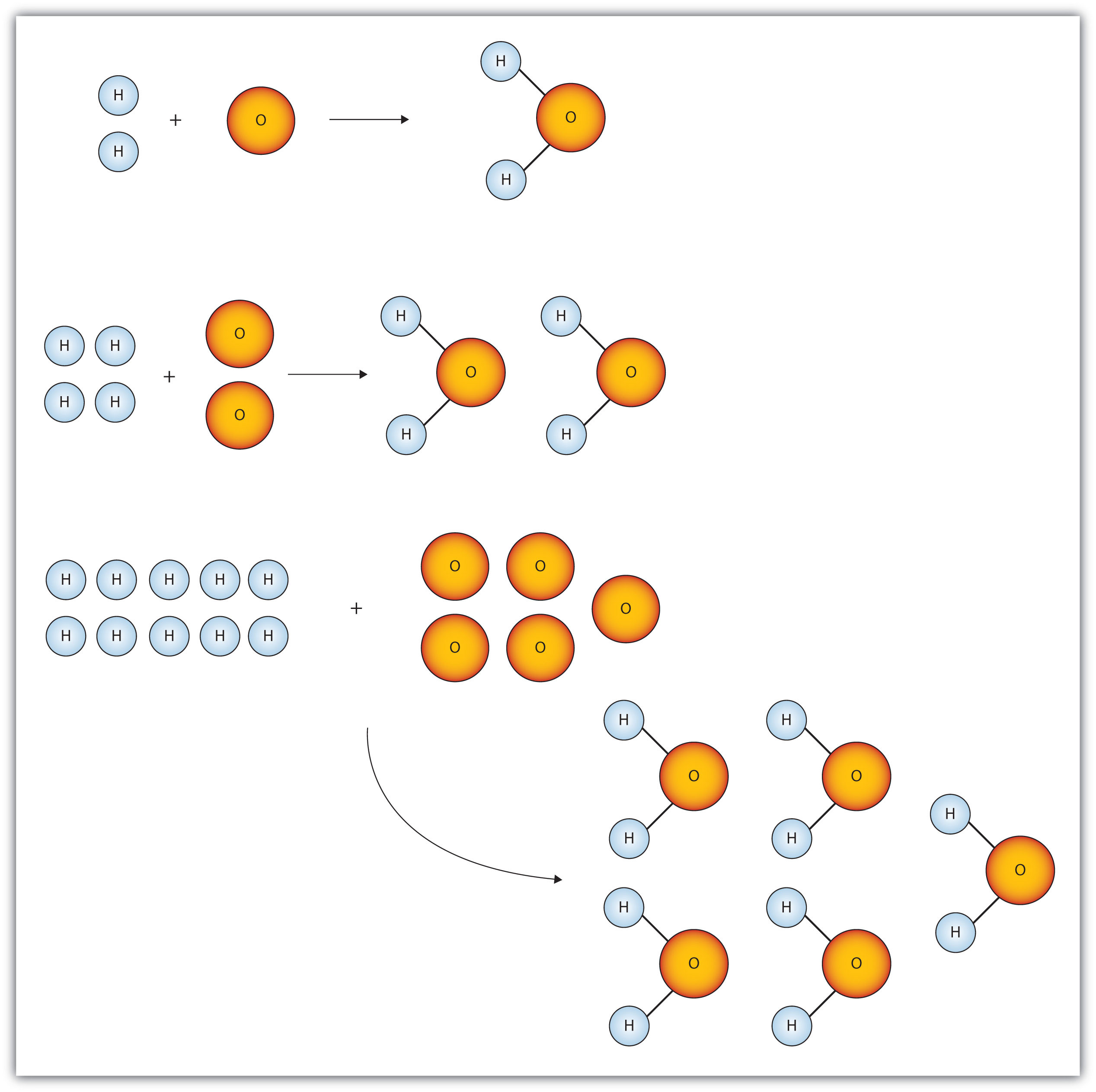
Chapter 6 Quantities In Chemical Reactions Chemistry

Chemical Reactions Anatomy And Physiology
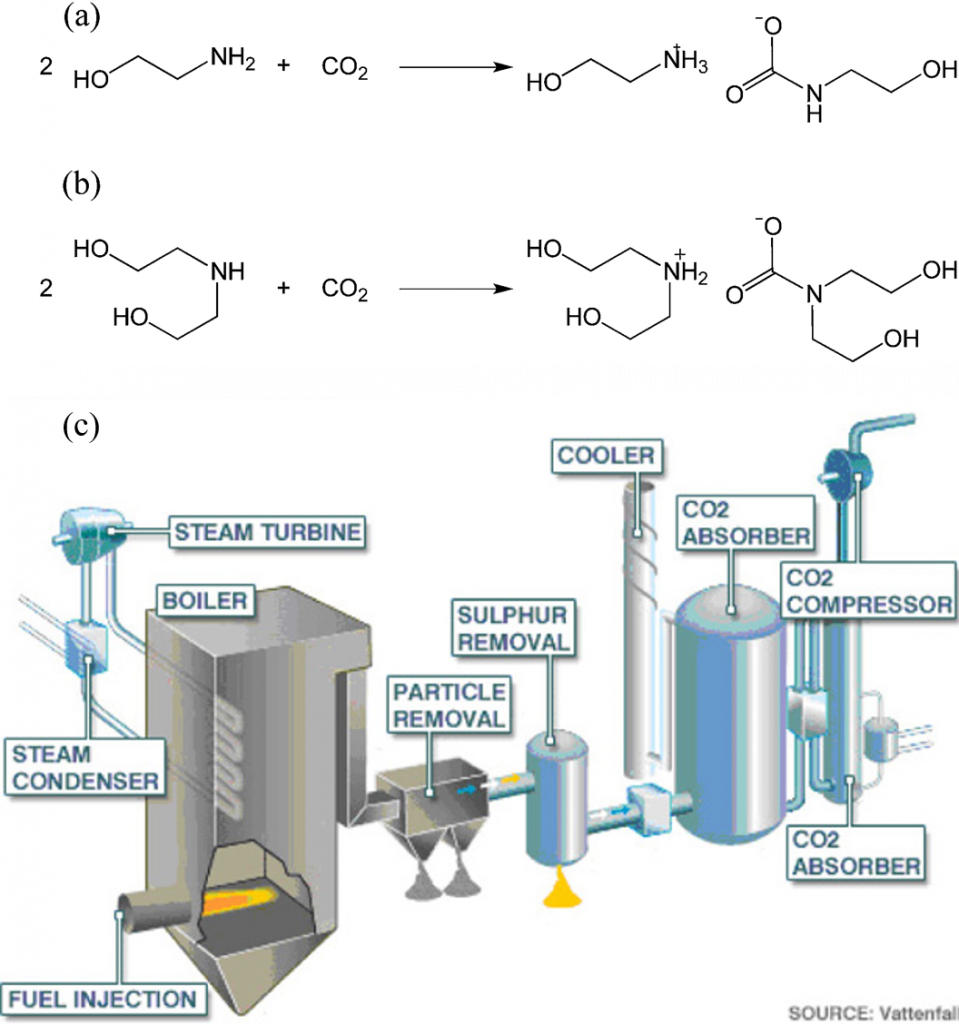
Ch104 Chapter 6 Quantities In Chemical Reactions Chemistry

Regrann From Chemicalacademy Different Kinds Of Reactions Organic Chemistry Teaching Chemistry Organic Chemistry Reactions

Chemical Reaction Millard Reaction Baking Bread Is A Millard Reaction Changes Color
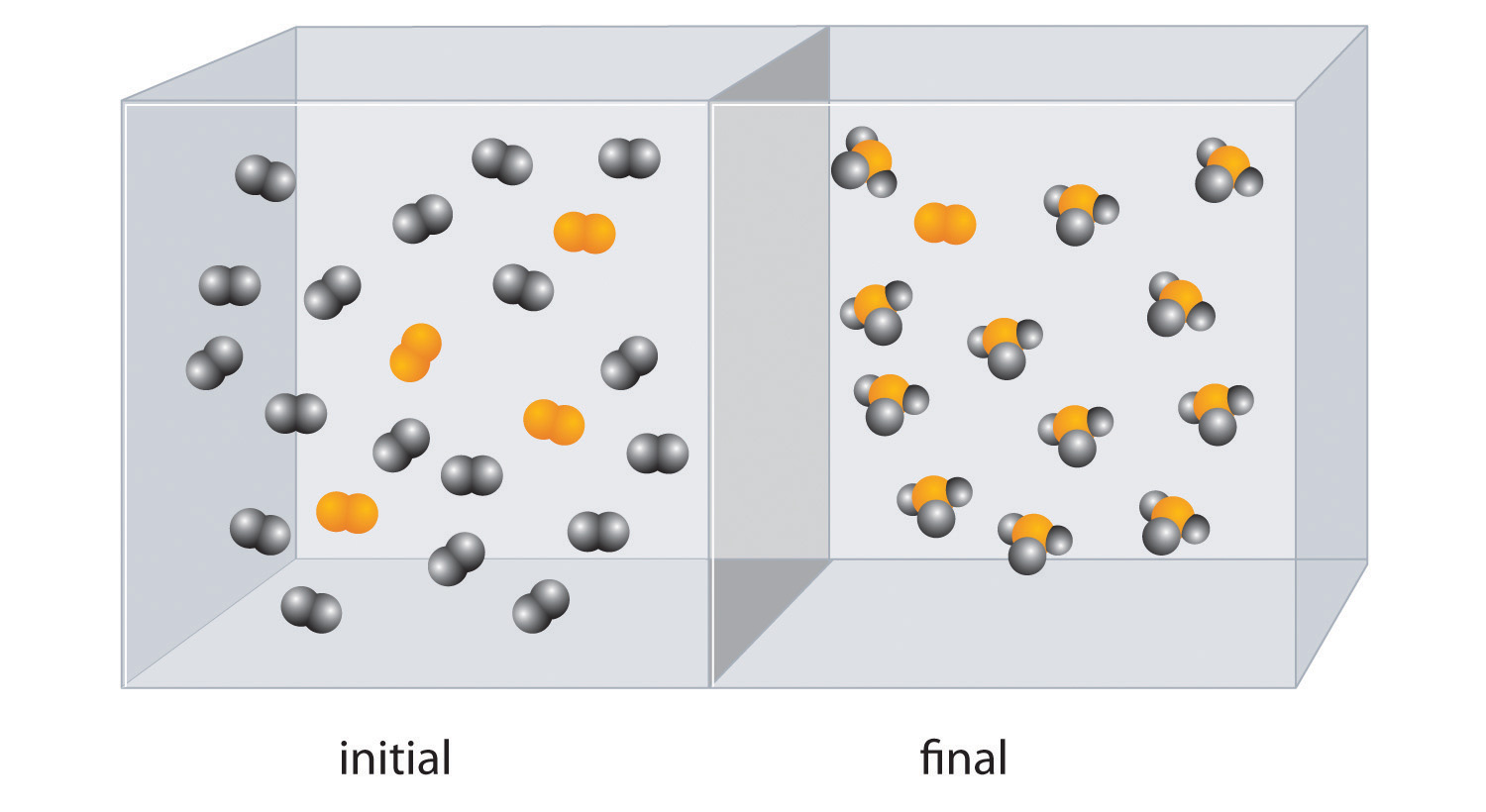
Classifying Chemical Reactions

Ch103 Chapter 7 Chemical Reactions In Biological Systems Chemistry
Comments
Post a Comment